 Open Nav
Open Nav
 Open Nav
Open Nav
Our research focuses on development of polymers for electron-transfer-based organic electronics, such as organic batteries and capacitors using redox polymers with high charge-storage densities. We have found that an ultimate population of redox sites allows efficient redox gradient-driven charge transport through self-exchange reactions. A new class of electroactive materials useful for wet-type energy storage, transport, and conversion devices is emerging. Electric field-driven charge transport is another remarkable aspect of these types of polymers in the solid state, with potential applications as organic memories, diodes, and switches.
Electrochemistry of redox polymers is dominated by charge transport processes based on electron self-exchange reactions and mass transfer processes of ions to maintain electroneutrality. Cyclic voltammogram of TEMPO-substituted polynorbornene (Figure) is approximated by an ideal surface wave expressed as i = n2F2vAΓ*ζ/(RT(1+ζ)2, where ζ = exp[(nF/RT)(E – E°)] and Γ* the total coverage of TEMPO, indicating that all the TEMPO units are equilibrated with the electrode potential. Such properties arise from the stability of both the oxidized and reduced states and efficient charge propagation across the polymer layer driven by the redox gradient. The electron self-exchange rate constants are exceptionally large for organic molecules, reflecting the large rate constant for the outer-sphere redox reaction of TEMPO. The charge propagating front obtained from the diffusion coefficient is useful for characterizing the redox polymers as charge transport materials in various wet-type devices.
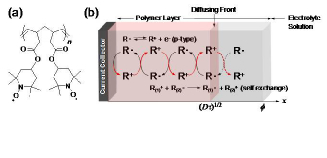
a) Typical redox polymer for charge transport and storage. (b) Charge propagation process.
B. Eng. (Waseda University 1990), Ph.D. (Waseda University 1995), JSPS Research Associate (1992-94), Visiting Research Associate, California Institute of Technology (1992-1994), Research Assistant, Department of Applied Chemistry, Waseda University (1995-96), Lecturer, Advanced Research Institute for Science and Engineering, Waseda University (1997-2003), Associate Professor, Institute of Colloid and Interface Science, Tokyo University of Science (2003-2006), Associate Professor, Faculty of Science and Engineering, Waseda University (2007-present). Award for Encouragement of Research in Polymer Science; The Society of Polymer Science, Japan (2001), Chemical Society of Japan Award for Young Chemists (2002).